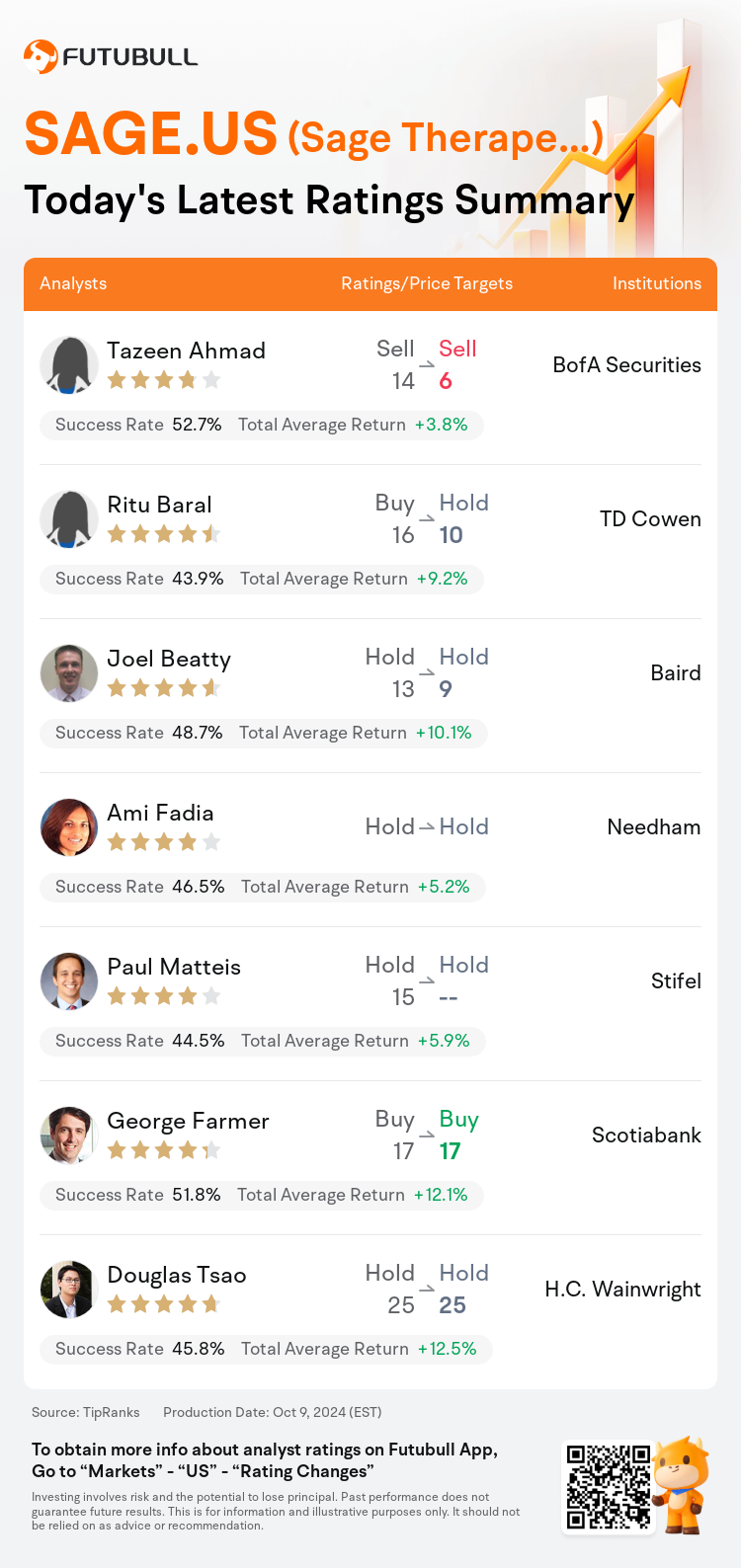
On Oct 09, major Wall Street analysts update their ratings for $Sage Therapeutics (SAGE.US)$, with price targets ranging from $6 to $25.
BofA Securities analyst Tazeen Ahmad maintains with a sell rating, and adjusts the target price from $14 to $6.
TD Cowen analyst Ritu Baral downgrades to a hold rating, and adjusts the target price from $16 to $10.
 Baird analyst Joel Beatty maintains with a hold rating, and adjusts the target price from $13 to $9.
Baird analyst Joel Beatty maintains with a hold rating, and adjusts the target price from $13 to $9.
Needham analyst Ami Fadia maintains with a hold rating.
Stifel analyst Paul Matteis maintains with a hold rating.
Furthermore, according to the comprehensive report, the opinions of $Sage Therapeutics (SAGE.US)$'s main analysts recently are as follows:
Following the Phase 2 LIGHTWAVE study results, where dalzanemdor did not meet the primary endpoint in Alzheimer's disease, and the subsequent announcement that development in this indication would not continue, the outlook on Sage Therapeutics has been impacted. Despite this setback, the company anticipates topline Phase 2 results from the DIMENSION trial assessing dalzanemdor in Huntington's disease later in the year. Initial positive findings from the SURVEYOR trials have provided some clinical substantiation; however, it's noted that substantial clinical proof is still required to mitigate risks associated with the program.
Sage Therapeutics' Phase 2 LIGHTWAVE study of dalzanemdor in mild Alzheimer's Disease/Mild Cognitive Impairment did not achieve a statistically significant improvement from the base measurement on the primary goal of WAIS-IV score change. It has been noted that this outcome was not factored into prior forecasts, therefore no adjustments are necessary. Sage is halting further development of dalzanemdor in Alzheimer's Disease, yet anticipates results from the DIMENSION study in Huntington's disease by the fourth quarter of 2024. Due to previous setbacks with dalzanemdor, there is a cautious stance regarding the likelihood of significant results in the upcoming Huntington's disease data readout.
Sage Therapeutics' recent announcement of the phase 2 LIGHTWAVE trial's failure of dalzanemdor in treating Alzheimer's disease has shifted the entirety of investor attention towards the prospects of Zurzuvae.
Here are the latest investment ratings and price targets for $Sage Therapeutics (SAGE.US)$ from 7 analysts:
Note:
TipRanks, an independent third party, provides analysis data from financial analysts and calculates the Average Returns and Success Rates of the analysts' recommendations. The information presented is not an investment recommendation and is intended for informational purposes only.
Success rate is the number of the analyst's successful ratings, divided by his/her total number of ratings over the past year. A successful rating is one based on if TipRanks' virtual portfolio earned a positive return from the stock. Total average return is the average rate of return that the TipRanks' virtual portfolio has earned over the past year. These portfolios are established based on the analyst's preliminary rating and are adjusted according to the changes in the rating.
TipRanks provides a ranking of each analyst up to 5 stars, which is representative of all recommendations from the analyst. An analyst's past performance is evaluated on a scale of 1 to 5 stars, with more stars indicating better performance. The star level is determined by his/her total success rate and average return.
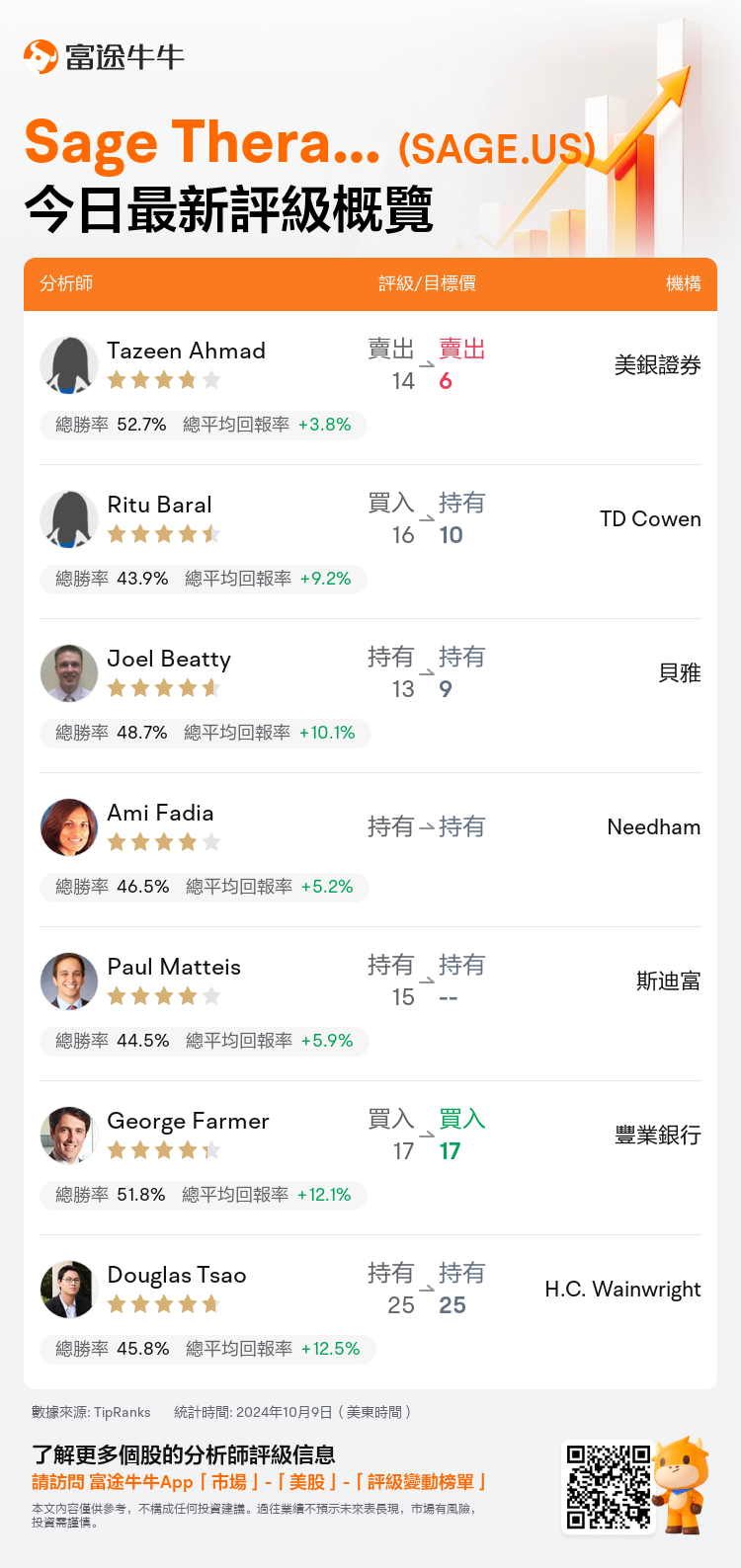
美東時間10月9日,多家華爾街大行更新了$Sage Therapeutics (SAGE.US)$的評級,目標價介於6美元至25美元。
美銀證券分析師Tazeen Ahmad維持賣出評級,並將目標價從14美元下調至6美元。
TD Cowen分析師Ritu Baral下調至持有評級,並將目標價從16美元下調至10美元。
 貝雅分析師Joel Beatty維持持有評級,並將目標價從13美元下調至9美元。
貝雅分析師Joel Beatty維持持有評級,並將目標價從13美元下調至9美元。
Needham分析師Ami Fadia維持持有評級。
斯迪富分析師Paul Matteis維持持有評級。
此外,綜合報道,$Sage Therapeutics (SAGE.US)$近期主要分析師觀點如下:
在2期LIGHTWAVE研究結果公佈後,達音那麥多爾未達到阿爾茨海默病的主要終點,並隨後宣佈該適應症的開發將不會繼續,對sage therapeutics的前景產生了影響。儘管遭遇挫折,該公司預計將在今年晚些時候從DIMENSION試驗中獲得2期初步結果,評估達音那麥多爾在亨廷頓病中的療效。來自SURVEYOR試驗的初步積極發現提供了一些臨床證實;然而,需要充分的臨床證據來減輕與該項目相關的風險。
sage therapeutics在輕度阿爾茨海默病/輕度認知障礙的達音那麥多爾2期LIGHTWAVE研究未能在主要目標WAIS-IV分數變化上實現統計學上的顯著改善。已經注意到先前的預測沒有考慮到這一結果,因此不需要進行調整。Sage正在停止在阿爾茨海默病中進一步開發達音那麥多爾,但預計將在2024年第四季度從DIMENSION研究中獲得有關亨廷頓病的結果。由於達音那麥多爾之前的挫折,對即將公佈的亨廷頓病數據持謹慎態度。
sage therapeutics最近宣佈2期LIGHTWAVE試驗中達音那麥多爾治療阿爾茨海默病失敗,導致投資者的全部注意力轉向Zurzuvae的前景。
以下爲今日7位分析師對$Sage Therapeutics (SAGE.US)$的最新投資評級及目標價:
提示:
TipRanks為獨立第三方,提供金融分析師的分析數據,並計算分析師推薦的平均回報率和勝率。提供的信息並非投資建議,僅供参考。本文不對評級數據和報告的完整性與準確性做出認可、聲明或保證。
TipRanks提供每位分析師的星級,分析師星級代表分析師所有推薦的過往表現,通過分析師的總勝率和平均回報率综合計算得出,星星越多,則該分析師過往表現越優異,最高爲5颗星。
分析師總勝率為近一年分析師的評級成功次數占總評級次數的比率。評级的成功與否,取決於TipRanks的虚擬投資組合是否從該股票中產生正回報。
總平均回報率為基於分析師的初始評級創建虚擬投資組合,並根據評級變化對組合進行調整,在近一年中該投資組合所獲得的回報率。
 貝雅分析師Joel Beatty維持持有評級,並將目標價從13美元下調至9美元。
貝雅分析師Joel Beatty維持持有評級,並將目標價從13美元下調至9美元。 
 Baird analyst Joel Beatty maintains with a hold rating, and adjusts the target price from $13 to $9.
Baird analyst Joel Beatty maintains with a hold rating, and adjusts the target price from $13 to $9.