Citi analyst David Lebowitz maintains $Intellia Therapeutics (NTLA.US)$ with a hold rating, and adjusts the target price from $29 to $19.
According to TipRanks data, the analyst has a success rate of 51.0% and a total average return of 1.4% over the past year.
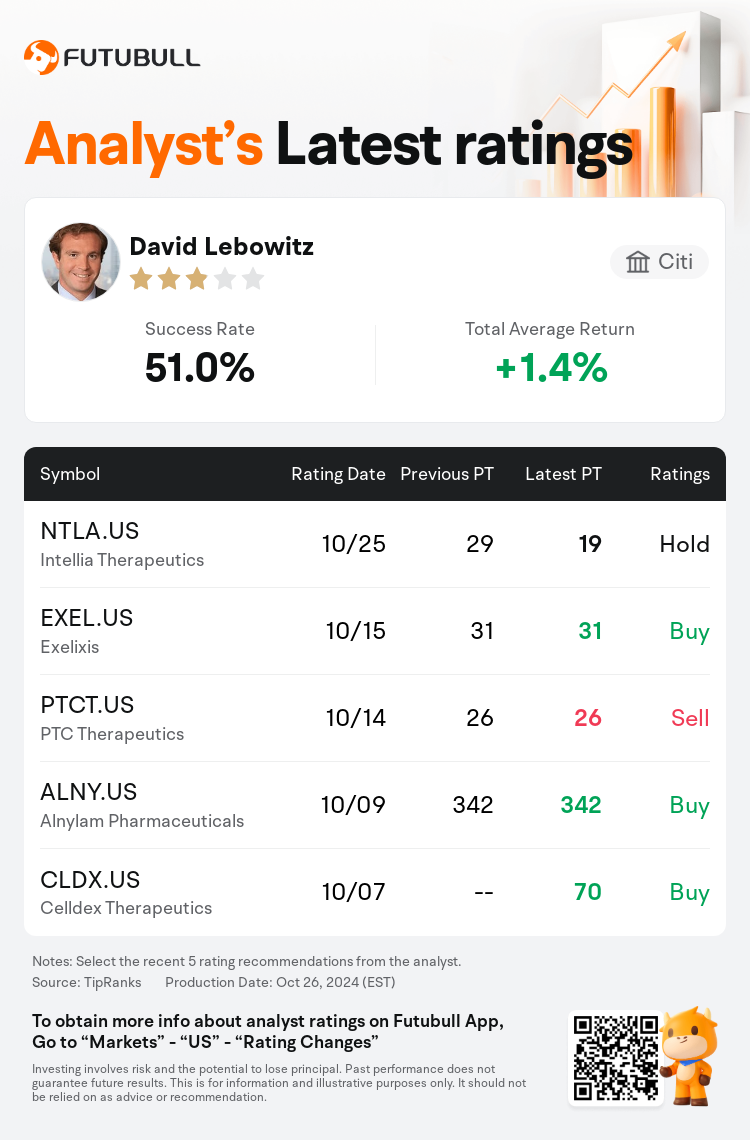
 Furthermore, according to the comprehensive report, the opinions of $Intellia Therapeutics (NTLA.US)$'s main analysts recently are as follows:
Furthermore, according to the comprehensive report, the opinions of $Intellia Therapeutics (NTLA.US)$'s main analysts recently are as follows:
The detailed data presented from the Phase 2 study of NTLA-2002 versus placebo in hereditary angioedema, or HAE, suggests gene editing could potentially offer a one-time 'functional cure.' However, the irreversible nature of gene editing sets a high threshold for market acceptance, given the unknown long-term safety implications of this method. It remains uncertain whether NTLA-2002's efficacy stands out compared to currently available treatments.
Recent updates on Intellia Therapeutics' NTLA-2002 Phase 2 data indicated a reduction in attacks, which seemed less impressive when compared to the initial Phase 1 results for HAE presented in June at EAACI. Despite the attack reduction being within a range that rivals that of competitors' Phase 3 trials, the distinctive 'one-and-done' treatment approach of NTLA-2002 is highlighted as an advantage over other treatments that require more frequent dosing. There remains a competitive market opportunity for NTLA-2002, although expectations for its peak market share have been moderated, along with a more cautious perspective on the probability of success for NTLA-2001 in ATTR, noting that additional validation of the platform is necessary.
The perspective on Intellia Therapeutics has become incrementally negative after the results of the Phase 2 study of NTLA-2002 in patients with hereditary angioedema (HAE).
Note:
TipRanks, an independent third party, provides analysis data from financial analysts and calculates the Average Returns and Success Rates of the analysts' recommendations. The information presented is not an investment recommendation and is intended for informational purposes only.
Success rate is the number of the analyst's successful ratings, divided by his/her total number of ratings over the past year. A successful rating is one based on if TipRanks' virtual portfolio earned a positive return from the stock. Total average return is the average rate of return that the TipRanks' virtual portfolio has earned over the past year. These portfolios are established based on the analyst's preliminary rating and are adjusted according to the changes in the rating.
TipRanks provides a ranking of each analyst up to 5 stars, which is representative of all recommendations from the analyst. An analyst's past performance is evaluated on a scale of 1 to 5 stars, with more stars indicating better performance. The star level is determined by his/her total success rate and average return.
花旗分析師David Lebowitz維持$Intellia Therapeutics (NTLA.US)$持有評級,並將目標價從29美元下調至19美元。
根據TipRanks數據顯示,該分析師近一年總勝率為51.0%,總平均回報率為1.4%。
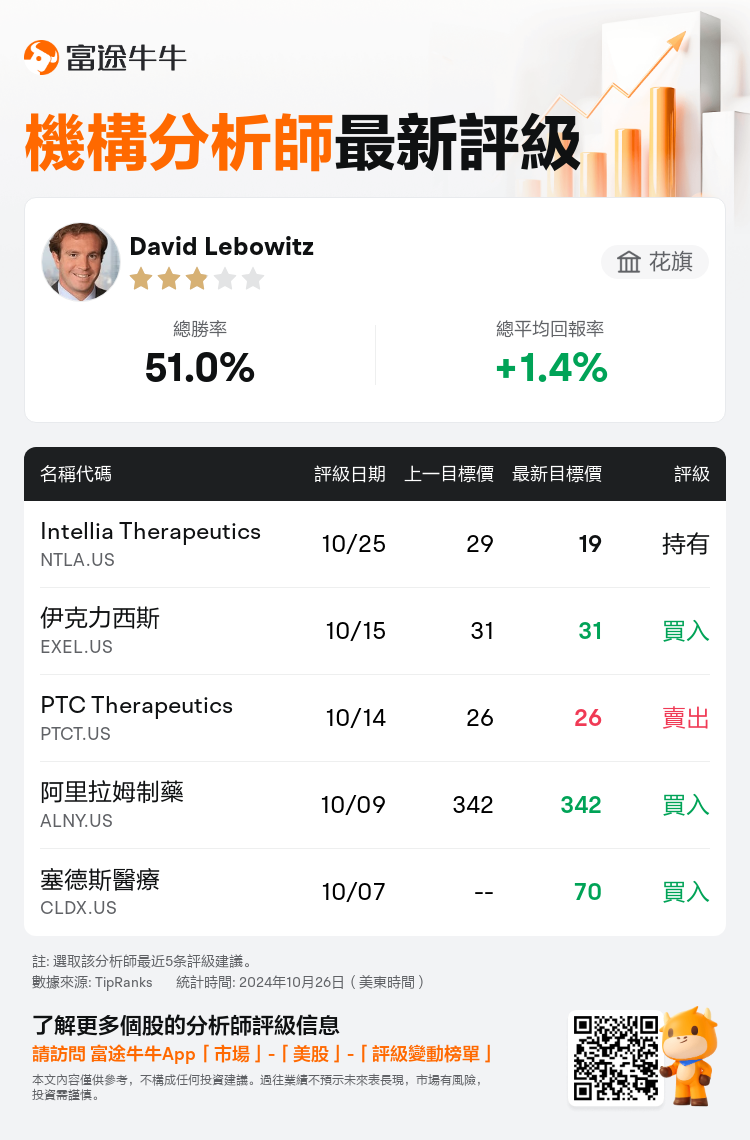
 此外,綜合報道,$Intellia Therapeutics (NTLA.US)$近期主要分析師觀點如下:
此外,綜合報道,$Intellia Therapeutics (NTLA.US)$近期主要分析師觀點如下:
NTLA-2002在遺傳性血管性水腫(HAE)的2期研究中呈現的詳細數據與安慰劑相比,基因編輯有可能提供一次性的'功能性治癒'。然而,基因編輯的不可逆性給市場接受設置了很高的門檻,考慮到這種方法的長期安全影響尚不明確。尚不清楚NTLA-2002的療效是否可以與當前可用的治療方法相媲美。
近期對Intellia Therapeutics的NTLA-2002第2期數據的更新表明,攻擊次數有所減少,但與6月份在EAACI展示的HAE的初始第1期結果相比,印象較差。儘管攻擊次數的減少處於可與競爭對手的第3期試驗相媲美的區間內,但NTLA-2002獨特的'一次性'治療方式被突出爲比其他需要更頻繁服藥的治療方案更有優勢。雖然NTLA-2002仍有競爭市場機會,但對其峯值市場份額的預期已有所調整,並對NTLA-2001在ATTR中的成功概率持更謹慎態度,指出有必要進一步驗證該平台。
隨着NTLA-2002在遺傳性血管性水腫(HAE)患者中的2期研究結果,對Intellia Therapeutics的展望逐漸變得負面。
提示:
TipRanks為獨立第三方,提供金融分析師的分析數據,並計算分析師推薦的平均回報率和勝率。提供的信息並非投資建議,僅供参考。本文不對評級數據和報告的完整性與準確性做出認可、聲明或保證。
TipRanks提供每位分析師的星級,分析師星級代表分析師所有推薦的過往表現,通過分析師的總勝率和平均回報率综合計算得出,星星越多,則該分析師過往表現越優異,最高爲5颗星。
分析師總勝率為近一年分析師的評級成功次數占總評級次數的比率。評级的成功與否,取決於TipRanks的虚擬投資組合是否從該股票中產生正回報。
總平均回報率為基於分析師的初始評級創建虚擬投資組合,並根據評級變化對組合進行調整,在近一年中該投資組合所獲得的回報率。
 此外,綜合報道,$Intellia Therapeutics (NTLA.US)$近期主要分析師觀點如下:
此外,綜合報道,$Intellia Therapeutics (NTLA.US)$近期主要分析師觀點如下:
 Furthermore, according to the comprehensive report, the opinions of
Furthermore, according to the comprehensive report, the opinions of